Updates and Corrections—October 2025 (Page 2)
Lumps
Imperial Cormorant Leucocarbo atriceps subsumes seven species
Summary: (7→1 species) The subantarctic shags or cormorants must have greater dispersal capabilities than it seems, judging by their recent divergence and plumage similarity.
Details: v2025 taxa 7446–7457
Was:
- South Georgia Shag Leucocarbo georgianus (monotypic)
- Antarctic Shag Leucocarbo bransfieldensis (monotypic)
- Crozet Shag Leucocarbo melanogenis (monotypic)
- Kerguelen Shag Leucocarbo verrucosus (monotypic)
- Heard Island Shag Leucocarbo nivalis (monotypic)
- Macquarie Shag Leucocarbo purpurascens (monotypic)
- Imperial Cormorant Leucocarbo atriceps
- subspecies atriceps and albiventer
Now:
- Imperial Cormorant Leucocarbo atriceps
- subspecies georgianus, bransfieldensis, melanogenis, verrucosus, nivalis, purpurascens, atriceps, and albiventer
Graphical abstract:
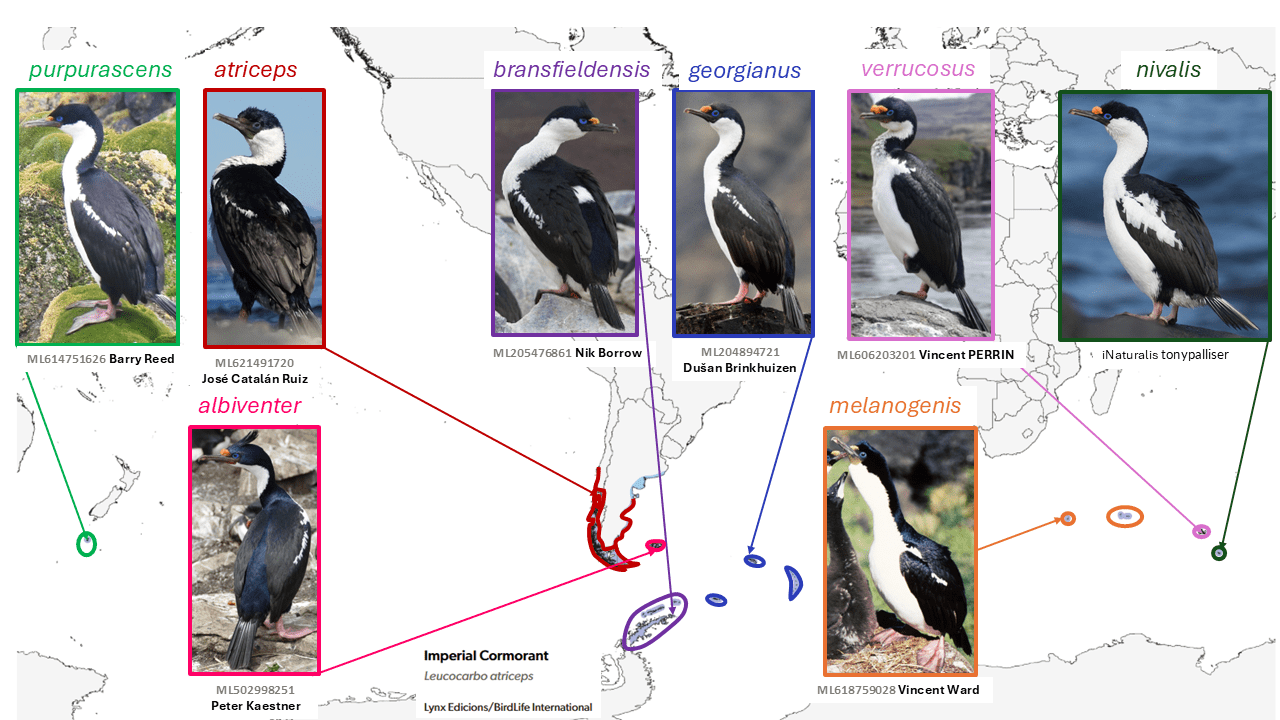
The subantarctic shags now in the genus Leucocarbo exhibit puzzling patterns of morphological variation over a vast and extraordinarily broadly allopatric range that have long challenged attempts at species-level taxonomy. For many years, most authors (including Peters 1931) treated them as three species: (1) Phalacrocorax atriceps (King, 1828), including atriceps of southwestern South America and the South Shetland Islands, and traversi Rothschild, 1898, now purpurascens (Brandt, 1837) of Macquarie Island (in the subantarctic between Australia and New Zealand); (2) Phalacrocorax albiventer (Lesson, 1831), including georgianus (Lönnberg, 1906) and vanhöffeni Reichenow, 1904, now melanogenis (Blyth, 1860) of the Crozet Islands, and (3) verrucosus (Cabanis, 1875) of Kerguelen. Two further forms in the complex were described after the appearance of Peters (1931): bransfieldensis (Murphy,1936) and nivalis (Falla, 1937). The long-standing Peters (1931) treatment, which united the two South Pacific taxa as atriceps, to the exclusion of the other taxa, is nevertheless untenable both in terms of morphology and biogeography, as atriceps and purpurascens differ strongly in cheek pattern and are separated by nearly 8000 km, while albiventer unites taxa virtually indistinguishable from nominate atriceps with others hardly differing from purpurascens.
While the two South American forms were earlier considered separate species (as in Peters 1931), Devillers and Terschuren (1978) showed that there appeared to be considerable intergradation in the Tierra del Fuego region between the dark-cheeked eastern form albiventer and the white-cheeked western atriceps. Further study of morphology and allozymes confirmed the lack of reproductive isolation between these two taxa (Rasmussen 1994), leading to their treatment as conspecific. However, Marchant and Higgins (1990) considered the complex to involve multiple species, though without a comprehensive published analysis. Siegel-Causey and Lefevre (1989) believed that the Antarctic form bransfieldensis was diagnosable on the basis of osteology and morphometrics, and that midden remains showed past sympatry. On this basis, a seven-species treatment was widely adopted, including the long-standing recognition of the Kerguelen form verrucosus as specifically distinct (following Voisin 1970).
All these shags are highly site-specific and rarely encountered far away from their breeding colonies. Perplexingly, the strongest morphological differences of all occur between the small verrucosus of Kerguelen (which has the greatest extent of black in adults and also has uniquely dark immatures) and the Heard Island form nivalis, which has the greatest extent of white on auriculars and wing of any form, although these two occupy islands separated by less than 500 km. Claims of diagnosability of Antarctic bransfieldensis and its sympatry with atriceps (Siegel-Causey and Lefevre 1989) have not been substantiated, and SACC (Remsen et al. 2025) consider them conspecific.
However, Calderon et al. (2014) found that Leucocarbo atriceps (including albiventer) shows little genetic structure relative to its more sedentary congener Magellanic Cormorant Leucocarbo magellanicus (Gmelin, 1789). Furthermore, a recent study shows that all the subantarctic taxa treated by Peters (1931) as comprising the three species atriceps, albiventer, and verrucosus, and including Antarctic bransfieldensis, are very closely related genetically (Rawlence et al. 2022). Thus they are all treated as conspecific by AviList Core Team (2025) and here. Genomic level work or comprehensive comparisons of behavior and vocalizations might however show previously undetected differentiation.
English names: The group names ‘shag’ and ‘cormorant’ have received approximately equally wide usage, and are essentially interchangeable, though some species have normally or always been referred to by one or the other name. In this group, the names ‘Blue-eyed Shag’ and ‘Imperial Cormorant’ have both been used extensively. Given that SACC (Remsen et al. 2025) has long used ‘Imperial Cormorant’ for Leucocarbo atriceps, that is the name used here for the enlarged species (although SACC retains Leucocarbo within Phalacrocorax).
Tawny Owl Strix aluco subsumes Maghreb Owl Strix mauritanica
Summary: (2→1 species) A North African owl is returned to subspecies status under the Tawny Owl, pending further study.
Details: v2025 taxa 9199–9208
Was:
- Maghreb Owl Strix mauritanica (monotypic)
- Tawny Owl Strix aluco
- subspecies aluco, siberiae, sylvatica, willkonskii, sanctinicolai, harmsi, and biddulphi
Now:
- Tawny Owl Strix aluco
- subspecies aluco, siberiae, sylvatica, willkonskii, sanctinicolai, harmsi, biddulphi, and mauritanica
Graphical abstract:
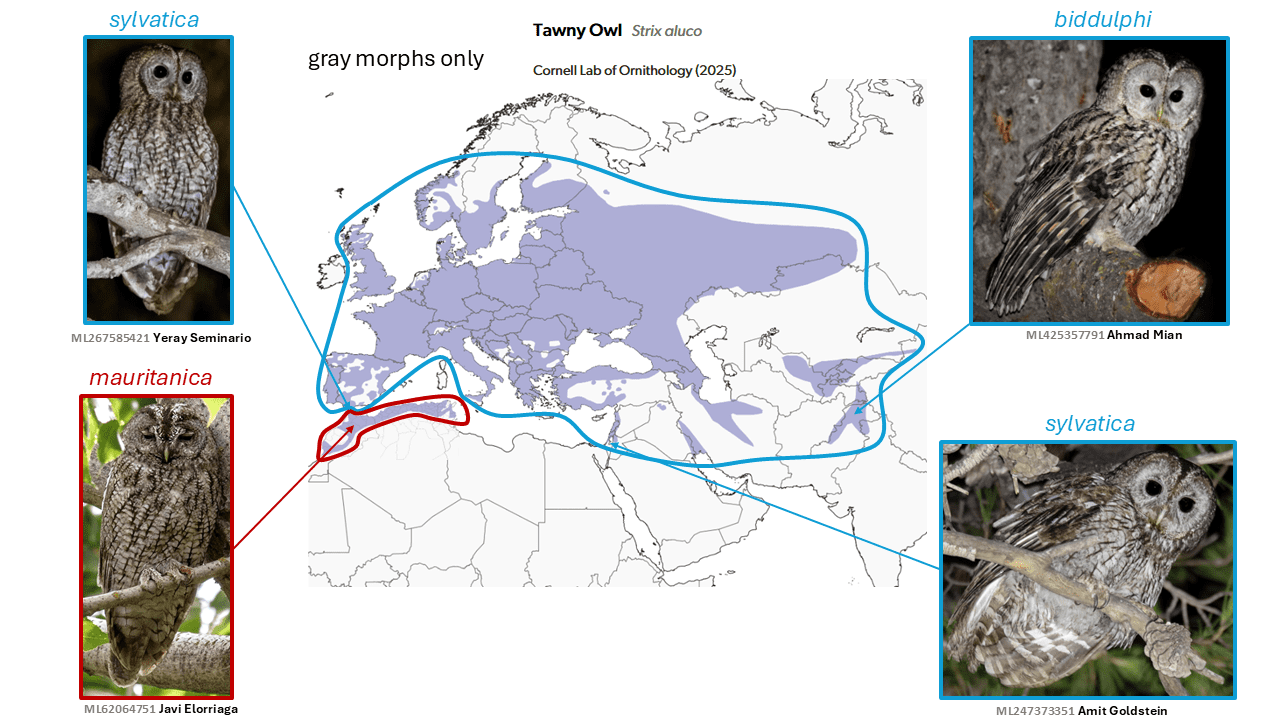
Originally described as a subspecies of Tawny Owl Strix aluco Linnaeus, 1758, and then not until 1905, taxon mauritanica (Witherby, 1905) was generally considered a subspecies (including by Peters 1940) until very recently. It was found to form a distinct lineage, though close to all other taxa of aluco sampled (Brito et al 2005, Doña et al. 2015), and the divergence age was estimated at around 500,000 ybp. Closer study of its morphology and vocalizations (Robb et al. 2015) led these authors to conclude that it is better treated as a full species. This argument is bolstered by its large size, contrary to the usual size cline, and abrupt morphological shift over the very narrow barrier formed by the Strait of Gibraltar. In addition to its large size, it has stronger cross-barring on the underparts and fairly dark plumage overall with no rufous morph (but the latter is also true of some eastern subspecies). Its vocalizations are broadly similar to those of other Tawny Owl subspecies, but notably deeper, and with subtle distinctions such as in pause length, number of notes, and harshness, and its excited pulsing hoot (Robb et al. 2015). These differences led IOC and eBird/Clements to adopt the recommended split.
However, Tawny Owls give notably variable vocalizations even in the same area, and no formal analysis has been performed, nor is it clear without playback experiments how the owls would perceive these rather subtle song differences. Given their overall similarity and the fairly recent divergence, mauritanica is considered by AviList Core Team (2025) to be a subspecies of Strix aluco.
English names: The name Tawny Owl is too deeply ingrained for any change to be imaginable, though the color ‘tawny’ does not describe the color of the newly lumped mauritanica.
Black-eared Catbird Ailuroedus melanotis subsumes five species
Summary: (5→1 species) A single-species treatment is adopted for five little-known taxa of catbird in New Guinea and far northern Australia, pending further study.
Details: v2025 taxa 17522–17532
Was:
- Huon Catbird Ailuroedus astigmaticus (monotypic)
- Black-capped Catbird Ailuroedus melanocephalus (monotypic)
- Northern Catbird Ailuroedus jobiensis (monotypic)
- Arfak Catbird Ailuroedus arfakianus
- subspecies misoliensis and arfakianus
- Black-eared Catbird Ailuroedus melanotis
- subspecies melanotis, facialis, and joanae
Now:
- Black-eared Catbird Ailuroedus melanotis
- subspecies melanotis, facialis, joanae, astigmaticus, melanocephalus, jobiensis, misoliensis, and arfakianus
Graphical abstract:

Map note: Taxon maculosa is here considered a separate species from the remaining taxa. Some range outline modifications follow Gregory (2025).
Formerly (as in Mayr 1962, in Volume 15 of the Peters’ Check-list) all treated as members of a single species, Ailuroedus crassirostris (Paykull, 1815), several of the taxa in the Ailuroedus melanotis (Gray, 1858) catbird complex were originally described as separate species, including melanotis, melanocephalus Ramsay, 1883, jobiensis Rothschild, 1895, and arfakianus Meyer, 1874. Recent phylogenetic work (Irestedt et al. 2016, Ericson et al. 2020) has demonstrated relatively deep divergences between all taxa, and this (despite phenotypic similarity of several taxa) led to their splitting by IOC-WBL and eBird/Clements.
Upon reconsideration by AviList, however, neither morphological nor vocal data appear to strongly bolster most aspects of the melanotis complex split based on genetics. The following taxa are now considered to be conspecific, under melanotis: melanotis, facialis, joanae, astigmaticus, melanocephalus, jobiensis, misoliensis, and arfakianus. These are placed in five new groups, corresponding to the previously recognized species, as summarized above.
The split between maculosus Ramsay, 1875 of northeastern Queensland (except the far north) and crassirostris is deeper, and they also differ more obviously in plumage from each other and from the melanotis groups; thus these are still considered full species in the taxonomy adopted by AviList Core Team (2025).
English names: As used by BirdLife Australia (2023), the name Black-eared Catbird is reverted to for the enlarged Ailuroedus melanotis complex, to the exclusion of Spotted Catbird Ailuroedus maculosus. However, it is acknowledged that this may lead to confusion, as a different treatment is adopted by e.g. Menkhorst et al. (2017), and further consideration is needed.
White-bellied Cuckooshrike Coracina papuensis subsumes Manus Cuckooshrike Coracina ingens
Summary: (2→1 species) Although the island of Manus, isolated northwest of New Britain, now loses one of its endemic species, the Manus Cuckooshrike, to lumping, further study may still validate its species status.
Details: v2025 taxa 18695–18710
Was:
- Manus Cuckooshrike Coracina ingens (monotypic)
- White-bellied Cuckooshrike Coracina papuensis
- subspecies papuensis, oriomo, angustifrons, louisiadensis, sclaterii, perpallida, elegans, eyerdami, timorlaoensis, hypoleuca, apsleyi, and artamoides
Now:
- White-bellied Cuckooshrike Coracina papuensis
- subspecies papuensis, oriomo, angustifrons, louisiadensis, ingens, sclaterii, perpallida, elegans, eyerdami, timorlaoensis, hypoleuca, apsleyi, and artamoides
Graphical abstract:

The Manus cuckooshrike taxon ingens (Rothschild & Hartert, 1914) was originally described as a subspecies of Coracina papuensis (Gmelin, 1788), and although the describers merely mentioned its larger size, the scientific name they bestowed on it translates to “huge”, and later they referred to it as ‘enormous’ (Rothschild and Hartert 1916). It was long treated as a subspecies, as by Mayr and Greenway (1960, in Volume 9 of Peters’ Check-list), but was considered a separate species by Schodde and Mason (1999), followed by Dickinson (2003), IOC-WBL since v1.0, and eBird/Clements since v.6.7.
The results of multiple genetic studies, taken together, throw considerable uncertainty over the relationships in the Coracina papuensis complex and certain other taxa not normally considered part of the complex, but ingens seems to be only moderately diverged in mtDNA (Fuchs et al. 2007, Smith and Filardi 2007, Jønsson et al. 2010, McCullough et al. 2022). Vocal and morphometric analyses, and perhaps playback experiments, may throw further light on this issue. Recordings are currently unavailable publicly, and in the meantime it is treated as a subspecies and monotypic group of Coracina papuensis by AviList Core Team (2025).
English names: The highly familiar name White-bellied Cuckooshrike (e.g. BirdLife Australia 2023, which however uses as the group name Cuckoo-shrike) is retained for the enlarged Coracina papuensis.
Little Shrikethrush Colluricincla megarhyncha subsumes seven species
Summary: (7→1 species) The wide-ranging Little Shrikethrush of northern Australia and New Guinea may well comprise multiple species, but further research is needed.
Details: v2025 taxa 19329–19362
Was:
- Variable Shrikethrush Colluricincla fortis
- subspecies neos, despecta, superflua, fortis, and trobriandi
- Waigeo Shrikethrush Colluricincla affinis (monotypic)
- Mamberamo Shrikethrush Colluricincla obscura
- subspecies obscura, idenburgi, and hybridus
- Sepik-Ramu Shrikethrush Colluricincla tappenbecki
- subspecies tappenbecki, maeandrina, and madaraszi
- Tagula Shrikethrush Colluricincla discolor (monotypic)
- Rufous Shrikethrush Colluricincla rufogaster
- subspecies goodsoni, aelptes, normani, griseata, synaptica, gouldii, and rufogaster
- Arafura Shrikethrush Colluricincla megarhyncha
- subspecies batantae, misoliensis, aruensis, megarhyncha, ferruginea, wuroi, palmeri, and parvula
Now:
- Little Shrikethrush Colluricincla megarhyncha
- subspecies neos, despecta, superflua, fortis, trobriandi, affinis, obscura, idenburgi, hybridus, tappenbecki, maeandrina, madaraszi, batantae, misoliensis, aruensis, megarhyncha, ferruginea, wuroi, palmeri, parvula, discolor, goodsoni, aelptes, normani, griseata, synaptica, gouldii, and rufogaster
Graphical abstract:

Seven taxa of Colluricincla from northern Australia and New Guinea that were each originally described as full species, megarhyncha (Quoy and Gaimard, 1832), affinis (Gray, 1862), obscura (Meyer, 1874), fortis (Gadow, 1883), discolor De Vis, 1890, rufogaster Gould, 1845, and tappenbecki Reichenow, 1899, were all treated for many years as members of a single species, Colluricincla megarhyncha, including by Mayr (1967, in Volume 12 of Peters’ Check-list).
The results of multiple genetic studies (Deiner et al. 2011, Marki et al. 2018, and Brady et al. 2022) led some authorities to treat the Collurincla megarhyncha complex as comprising seven species, as outlined above. However, issues with paraphyly and apparent intergradation cloud the picture, and pending further research they are all considered groups within the highly polytypic C. megarhyncha by AviList Core Team (2025).
English names: The long-used English name Little Shrikethrush Colluricincla megarhyncha (as in BirdLife Australia 2023, which however formats the group name as Shrike-thrush) is reverted to here.
Generic Changes
Resurrect Megaloprepia for two species of Ptilinopus
Summary: (1→2 genera; see following accounts for Ramphiculus and Drepanoptila)
Details: Clements v2025 taxa 2585–2593
Was:
- Ptilinopus bernsteinii
- subspecies bernsteinii and micrus
- Ptilinopus magnificus
- subspecies puella, poliurus, assimilis, keri, and magnificus
Now:
- Megaloprepia formosa
- subspecies formosa and micra
- Megaloprepia magnifica
- subspecies puella, poliura, assimilis, keri, and magnifica
Based on the phylogenies of Cibois et al. (2014) and Nash et al. (2024), the broad fruit-dove genus Ptilinopus as recognized in Clements et al. (2024) and previous versions was shown to be non-monophyletic. Thus, two genera, Megaloprepia Reichenbach, 1853 (of the Moluccas, New Guinea, and Australia) and Ramphiculus Bonaparte, 1854 (of the Philippines, Malay Peninsula, Greater Sundas, and Sulawesi region) were resurrected by del Hoyo and Collar (2014), and adopted by AviList Core Team (2025) and Clements et al. (2025). Without benefit of a molecular phylogeny, Wolters (1975) treated most of these and several others in the genus Megaloprepia, but placed species other than magnifica and formosa in different subgenera (including Ramphiculus). See Sequence Changes section for new linear sequence.With the genus change, the specific epithet of one species that had been changed due to preoccupation (Mees 1973, Dickinson and Remsen 2013) reverts from Ptilinopus bernsteinii to Megaloprepia formosa, as the name formosa is no longer preoccupied (del Hoyo and Collar 2014).
Resurrect Ramphiculus for nine species of Ptilinopus
Summary: (1→2 genera; see previous account for Megaloprepia and subsequent one for Drepanoptila)
Details: Clements v2025 taxa 2594–2614
Was:
- Ptilinopus marchei (monotypic)
- Ptilinopus merrilli
- subspecies faustinoi and merrilli
- Ptilinopus occipitalis
- subspecies occipitalis and incognitus
- Ptilinopus fischeri
- subspecies fischeri, centralis, and meridionalis
- Ptilinopus jambu (monotypic)
- Ptilinopus leclancheri
- subspecies taiwanus, longialis, leclancheri, and gironieri
- Ptilinopus mangoliensis (monotypic)
- Ptilinopus gularis (monotypic)
- Ptilinopus subgularis (monotypic)
Now:
- Ramphiculus marchei (monotypic)
- Ramphiculus merrilli
- subspecies faustinoi and merrilli
- Ramphiculus occipitalis
- subspecies occipitalis and incognitus
- Ramphiculus fischeri
- subspecies fischeri, centralis, and meridionalis
- Ramphiculus jambu (monotypic)
- Ramphiculus leclancheri
- subspecies taiwanus, longialis, leclancheri, and gironieri
- Ramphiculus mangoliensis (monotypic)
- Ramphiculus gularis (monotypic)
- Ramphiculus subgularis (monotypic)
See previous account (Megaloprepia) for rationale behind this generic change.
Subsume Drepanoptila into Ptilinopus
Summary: (1→2 genera; see previous accounts for Megaloprepia and Ramphiculus)
Details: Clements v2025 taxon 2634
Was:
- Drepanoptila holosericea (monotypic)
Now:
- Ptilinopus holosericea (monotypic)
In the phylogenies of Cibois et al. (2014) and Nash et al. (2024), the monotypic New Caledonian genus Drepanoptila Bonaparte, 1855 (Cloven-feathered Dove) is embedded within Ptilinopus, but its exact position is poorly supported. AviList Core Team (2025) has therefore placed Drepanoptila within Ptilinopus, followed by Clements et al. (2025). Wolters (1975) retained Drepanoptila as a monotypic genus, as he did for Fijian Chrysoenas Bonaparte, 1854, and placed them far apart in his linear sequence.
Subsume Macropsalis into Hydropsalis
Summary: (2→1 genera)
Details: Clements v2025 taxon 3576
Was:
- Macropsalis forcipata (monotypic)
Now:
- Hydropsalis forcipata (monotypic)
A recent revision of a diverse clade of Neotropical nightjars (Costa et al. 2023) has led to a much better understanding of the complex relationships of these taxa. In addition to several generic-level changes enacted in 2024, the taxon remaining in Macropsalis Sclater, 1866, M. forcipata (Long-trained Nightjar of southeastern Brazil and northeastern Argentina), which was retained in a monotypic Macropsalis by Wolters (1976), is now transferred to Hydropsalis Wagler, 1832, aligning with AviList Core Team (2025).
Subsume Bugeranus and Anthropoides into Grus
Summary: (3→1 genera)
Details: Clements v2025 taxa 5712–5714
Was:
- Bugeranus carunculatus (monotypic)
- Anthropoides virgo (monotypic)
- Anthropoides paradiseus (monotypic)
Now:
- Grus carunculatus (monotypic)
- Grus virgo (monotypic)
- Grus paradiseus (monotypic)
According to the mtDNA phylogeny of Krajewsky et al. (2010), the single species in Bugeranus Gloger, 1842 (the Wattled Crane of Ethiopia) and the two in Anthropoides Vieillot, 1816 (Demoiselle and Blue cranes of the Palearctic and southern Africa, respectively) are sister to the core Grus Brisson, 1760 clade (which is Holarctic). They could either be kept as separate genera, with divergence times estimated as Miocene, or subsumed within Grus. Wolters (1975) treated them as three genera, Bugeranus, Tetrapteryx Thunberg, 1818 (for paradiseus), and Anthropoides(for virgo). AviList Core Team (2025) has adopted the single-genus approach, and thus an expanded Grus. However, a genomic phylogeny of Gruidae is needed to better resolve the relationships within this ancient family.
Resurrect Hesperiphona for Coccothraustes vespertina and Coccothraustes abeillei
Summary: (1→2 genera)
Details: Clements v2025 taxa 32080–32090
Was:
- Coccothraustes vespertinus
- subspecies brooksi, vespertinus, and montanus
- Coccothraustes abeillei
- subspecies pallidus, saturatus, abeillei, and cobanensis
Now:
- Hesperiphona vespertina
- subspecies brooksi, californica, warreni, vespertina, and montana
- Hesperiphona abeillei
- subspecies pallida, saturata, abeillei, and cobanensis
The DNA-based phylogeny of Zuccon et al. (2012) strongly suggests that the Holarctic hawfinch/grosbeak genus Coccothraustes Brisson, 1760, as it has long been recognized by NACC and Clements, among others, is non-monophyletic with respect to Asian Mycerobas Cabanis, 1847 (Mason 2024, 2024-A-12, https://americanornithology.org/wp-content/uploads/2024/01/2024-A.pdf). One solution would be to lump Mycerobas into Coccothraustes, but the genus thus created would likely be far more deeply diverged than some others in the larger clade. Resurrection of the largely Nearctic genus Hesperiphona Bonaparte, 1850, as used by Wolters (1979) and others, for Evening Grosbeak and Hooded Grosbeak was therefore adopted by AviList Core Team (2025) and Clements et al. (2025). The above proposal did not pass NACC, however, due to the rather poor support for the relevant branches of the tree in Zuccon et al. (2012), and the lack of sampling of abeillei.
Resurrect Euthlypis for Basileuterus lachrymosus
Summary: (1→2 genera)
Details: Clements v2025 taxon 34224
Was:
- Basileuterus lachrymosus (monotypic)
Now:
- Euthlypis lachrymosa (monotypic)
In a recent UCE phylogeny (Zhao et al. 2025), the Fan-tailed Warbler Basileuterus lachrymosus of Mexico to northern Central America was recovered as sister to a large clade containing Basileuterus Cabanis, 1848, Cardellina Bonaparte, 1850, and Myioborus Baird, 1865, with 100% bootstrap support (summarized in Jiménez 2025, 2025-C-5). The species in question is an outlier from other species of Basileuterus in multiple ways, and rather than lump this highly heterogeneous clade, the name Euthlypis Cabanis, 1853, as used by Wolters (1980) and others, was resurrected by NACC (Chesser et al. 2025), and followed by Clements et al. (2025). AviList v.2026 is also expected to use Euthlypis.
Resurrect Leuconotopicus for six species of Dryobates
Summary: (1→3 genera; see also Veniliornis, below)
Details: Clements v2025 taxa 11426–11465
Was:
- Dryobates borealis (monotypic)
- Dryobates villosus
- subspecies septentrionalis, villosus, audubonii, terraenovae, piger, maynardi, sitkensis, picoideus, harrisi, hyloscopus, orius, icastus, jardinii, sanctorum, and extimus
- Dryobates albolarvatus
- subspecies albolarvatus and gravirostris
- Dryobates fumigatus
- subspecies oleagineus, sanguinolentus, fumigatus, obscuratus, and reichenbachi
- Dryobates stricklandi (monotypic)
- Dryobates arizonae
- subspecies arizonae and fraterculus
Now:
- Leuconotopicus borealis (monotypic)
- Leuconotopicus villosus
- subspecies septentrionalis, villosus, audubonii, terraenovae, piger, maynardi, sitkensis, picoideus, harrisi, hyloscopus, orius, icastus, jardinii, sanctorum, and extimus
- Leuconotopicus albolarvatus
- subspecies albolarvatus and gravirostris
- Leuconotopicus fumigatus
- subspecies oleagineus, sanguinolentus, fumigatus, obscuratus, and reichenbachi
- Leuconotopicus stricklandi (monotypic)
- Leuconotopicus arizonae
- subspecies arizonae and fraterculus
Based on phylogenetic studies that show three clades within the woodpecker genus Dryobates Boie, 1826 (mainly Shakya et al. 2017), three genera are recognized in this assemblage. In addition to Dryobates, which is now restricted to a Holarctic clade, AviList Core Team (2025) and now Clements et al. (2025) recognize these other two clades as the genus Leuconotopicus Malherbe, 1845, which is mainly Nearctic, and Veniliornis Bonaparte, 1854, which is exclusively Neotropical. Wolters (1976), without benefit of molecular phylogenies, also recognized Leuconotopicus, but for only two of the species in that clade, and Veniliornis, with a similar but not identical composition to that adopted here.
Resurrect Veniliornis for 14 species of Dryobates
Summary: (1→3 genera; see also Leuconotopicus, above)
Details: Clements v2025 taxa 11467–11514
Was:
- Dryobates kirkii
- subspecies neglectus, cecilii, kirkii, continentalis, and monticola
- Dryobates cassini (monotypic)
- Dryobates spilogaster (monotypic)
- Dryobates mixtus
- subspecies mixtus, berlepschi, malleator, and cancellatus
- Dryobates lignarius
- subspecies puncticeps and lignarius
- Dryobates sanguineus (monotypic)
- Dryobates passerinus
- subspecies fidelis, modestus, passerinus, diversus, agilis, olivinus, taenionotus, tapajozensis, and insignis
- Dryobates frontalis (monotypic)
- Dryobates callonotus
- subspecies callonotus and major
- Dryobates dignus
- subspecies dignus, baezae, and valdizani
- Dryobates nigriceps
- subspecies equifasciatus, pectoralis, and nigriceps
- Dryobates affinis
- subspecies orenocensis, hilaris, ruficeps, and affinis
- Dryobates chocoensis (monotypic)
- Dryobates maculifrons (monotypic)
Now:
- Veniliornis kirkii
- subspecies neglectus, cecilii, kirkii, continentalis, and monticola
- Veniliornis cassini (monotypic)
- Veniliornis spilogaster (monotypic)
- Veniliornis mixtus
- subspecies mixtus, berlepschi, malleator, and cancellatus
- Veniliornis lignarius
- subspecies puncticeps and lignarius
- Veniliornis sanguineus (monotypic)
- Veniliornis passerinus
- subspecies fidelis, modestus, passerinus, diversus, agilis, olivinus, taenionotus, tapajozensis, and insignis
- Veniliornis frontalis (monotypic)
- Veniliornis callonotus
- subspecies callonotus and major
- Veniliornis dignus
- subspecies dignus, baezae, and valdizani
- Veniliornis nigriceps
- subspecies equifasciatus, pectoralis, and nigriceps
- Veniliornis affinis
- subspecies orenocensis, hilaris, ruficeps, and affinis
- Veniliornis chocoensis (monotypic)
- Veniliornis maculifrons (monotypic)
See Leuconotopicus (above) for rationale for change.
Move Lophopsittacus bensoni to Psittacula, as Psittacula bensoni
Summary: (2→2 genera)
Details: Clements v2025 taxon 12388
Was:
- Lophopsittacus bensoni (monotypic)
Now:
- Psittacula bensoni (monotypic)
The extinct Mascarene psittacine Lophopsittacus bensoni Holyoak, 1973 was originally considered congeneric with the much larger Broad-billed Parrot Lophopsittacus mauritianus (Owen, 1866), formerly of Mauritius. However, Hume (2007) showed that bensoni is better placed in Psittacula, thus leaving only mauritianus in Lophopsittacus (see also https://en.wikipedia.org/wiki/Mascarene_grey_parakeet). This treatment was followed by the IOC-WBL, and subsequently by AviList Core Team (2025) and Clements et al. (2025).
Recognize Protopelma for Neopelma chrysolophum
Summary: (1→2 genera)
Details: Clements v2025 taxon 15694
Was:
- Neopelma chrysolophum (monotypic)
Now:
- Protopelma chrysolophum (monotypic)
The tyrant-manakins, subfamily Neopelminae, are notorious for their cryptic coloration in both sexes, in marked contrast to most other manakins Pipridae. The Serra do Mar Tyrant-Manakin Neopelma chrysolophum Pinto, 1944 of southeastern Brazil’s Atlantic forest, which was originally described as a subspecies of Neopelma aurifrons (Wied-Neuwied, 1831), and was included as such by Wolters (1977), has been shown to form a deeply divergent, monotypic clade (van Els et al. 2023). As no genus was available for this taxon, a new genus has been erected, Protopelma van Els et al., 2023, and a proposal to SACC (Remsen 2024) has passed and is thus enacted in Clements et al. (2023). AviList has also recently voted to recognize Protopelma, and this change is expected to appear in AviList Core Team (2026).
Nomenclature Changes
Change Ptilinopus bernsteinii to Megaloprepia formosa
Summary: (1→1 species)
Details: Clements v2025 taxa 2585–2587
Was:
- Ptilinopus bernsteinii
Now:
- Megaloprepia formosa
With the change in genus from Ptilinopus, the name bernsteinii is no longer preoccupied and thus the scientific name for Scarlet-breasted Fruit-Dove reverts to Megaloprepia formosa (del Hoyo and Collar 2014); see Generic Changes for more details.
Change Butorides atricapilla didi to Butorides atricapilla didii
Summary: (1→1 subspecies)
Details: Clements v2025 taxon 7684
Was:
- Butorides atricapilla didi
Now:
- Butorides atricapilla didii
The original spelling of the northern and central Maldives subspecies of Little Heron Butorides atricapilla is didii Phillips & Sims, 1958, rather than didi as often cited, as for example by Dickinson and Remsen (2013) and most subsequent sources, including previous versions of the Clements checklist (but spelled didii by Rasmussen and Anderton 2005). Article 33.4 (ICZN 1999) indicates that the original spelling must be used in this case.
This subspecies is often synonymized with Butorides atricapilla albolimbata Reichenow, 1900, of the Chagos Archipelago, as for example by Payne (1979, in Volume 1 Edition 2 of the Peters’ Check-list), followed by IOC-WBL and AviList Core Team (2025). However, a high percentage of individuals of didii are partially leucistic (see example photos which include many different individuals, but multiple images of some of them), unlike those of albolimbata, or of albidula Bangs, 1913 of the southern Maldives. Thus, pending investigation of the genetic structure of these taxa, Clements et al. (2025) continues to recognize didii (and albidula).
Correct Laterallus spilopterus to Laterallus spiloptera
Summary: (1→1 subspecies)
Details: Clements v2025 taxon 5659
Was:
- Laterallus spilopterus
Now:
- Laterallus spiloptera
The name spiloptera (Durnford, 1877) for Dot-winged Crake is treated as invariable, and thus is corrected from the masculine spilopterus to the original spelling, spiloptera.
Correct Nannopsittacus melanogenia suavissima to Nannopsittacus melanogenia suavissimus
Summary: (1→1 subspecies)
Details: Clements v2025 taxon 12523
Was:
- Nannopsittacus melanogenia suavissima
Now:
- Nannopsittacus melanogenia suavissimus
The specific epithet suavissimus Salvadori, 1876 is used instead of the feminine suavissima for Dusky-cheeked Fig-Parrot Nannopsittacus melanogenia, as the genus Nannopsittacus is masculine (Clements et al. 2025). See also discussion in Zoonomen and two Richmond Index cards (card one, card two) therein.
Change Family Scotocercidae to Family Cettiidae
Summary: (1→1 family)
Details: Clements v2025 taxa 25064–25172
Was:
- Scotocercidae
Now:
- Cettiidae
The family name Scotocercidae used in Fregin et al. (2012) and subsequent works is replaced by Cettiidae, which was made available by Coues (1903) and Chigi (1912). See Winkler et al. (2025) for further details.
Correct Arachnothera longirostra cinireicollis to Arachnothera longirostra cinereicollis
Summary: (1→1 subspecies)
Details: Clements v2025 taxon 30610
Was:
- Arachnothera longirostra cinireicollis
Now:
- Arachnothera longirostra cinereicollis
The longstanding original spelling cinireicollis (Latham, 1790), used by e.g. Rand (1967, in Volume 12 of Peters’ Check-list), Dickinson and Remsen (2014), and many others, is considered to be a misspelt word (e.g. del Hoyo and Collar 2016, N. David pers. comm. 2021), and was previously corrected (e.g. Chasen 1935, Wolters 1979). When considered an inadvertent error, as here, it must be corrected (ICZN 1999).
Correct Emblema modesta to Emblema modestum
Summary: (1→1 subspecies)
Details: Clements v2025 taxon 31031
Was:
- Emblema modesta
Now:
- Emblema modestum
The gender ending of the specific epithet Plum-headed Finch is changed to match the neuter gender of Emblema. Thus, Emblema modesta (Gould, 1837) becomes Emblema modestum.
Subspecies Changes
Resurrect Bostrychia hagedash erlangeri
Summary: (3→4 subspecies)
Details: Clements v2024 taxa 7516–7521
Was:
- Bostrychia hagedash brevirostris (part)
Now:
- Bostrychia hagedash erlangeri
Hadada Ibis Bostrychia hagedash erlangeri (Neumann, 1909), a split from subspecies brevirostris (Reichenow, 1907), is newly recognized. See under Changes to Groups for more details.
Recognize Megascops watsonii stangiae and Megascops watsonii ater
Summary: (2→4 subspecies)
Details: Clements v2024 taxa 8880–8884
Was:
- Tawny-bellied Screech-Owl Megascops watsonii
- subspecies watsonii and usta
Now:
- Tawny-bellied Screech-Owl Megascops watsonii
- subspecies watsonii, stangiae, ater, and usta
The integrative analysis of Dantas et al. (2021) has shown that Megascops watsonii is non-monophyletic with respect to Megascops atricapilla, and the Brazilian Ornithological Records Committee considers watsonii s.l. to be comprised of three species. Further study and adjustment of taxon limits will be necessary, but the distribution range of subspecies usta and watsonii have been modified in eBird/Clements based on Dantas et al. (2021). The two subspecies resurrected, stangiae Dantas et al., 2021 and ater (Hekstra, 1982), are in accord with Dantas et al. (2021). However, several aspects of the taxonomy of this complex remain unclear, especially regarding morphology and vocalizations, and more study is needed (see this SACC proposal, which did not pass, and Holt et al. 2025). See also Changes to Groups.
Resurrect Camptostoma imberbe imberbe and Camptostoma imberbe thyellophilum
Summary: (0→2 subspecies)
Details: Clements v2024 taxa 16551–16553
Was:
- Northern Beardless-Tyrannulet Camptostoma imberbe (monotypic)
Now:
- Northern Beardless-Tyrannulet (Northern) Camptostoma imberbe imberbe (monotypic)
- Northern Beardless-Tyrannulet (Yucatan) Camptostoma imberbe thyellophilum (monotypic)
The taxon thyellophilum Parkes & Phillips, 1999 of Northern Beardless Tyrannulet Camptostoma imberbe Sclater, 1857 has been synonymized by many, at least since the review of Fitzpatrick (2004). However, thyellophilum has recently been shown to be vocally if not morphologically distinctive (Lima and Vaz 2024). Although proposal 2025-B-01 to NACC did not pass, the recognition of this taxon is necessary, and it is also now considered a group (see Changes to Groups).
Resurrect Clytorhynchus nigrogularis nigrogularis and Clytorhynchus nigrogularis maximus
Summary: (0→2 subspecies)
Details: Clements v2024 taxa 20837–20839
Was:
- Clytorhynchus nigrogularis (monotypic)
Now:
- Clytorhynchus nigrogularis nigrogularis
- Clytorhynchus nigrogularis maximus
Long considered monotypic, the previously described Kadavu subspecies maximus Layard, 1876 of Black-throated Shrikebill Clytorhynchus nigrogularis Layard, 1875 is resurrected, stimulated by recent observations and media that affirm its distinctiveness (Gregory et al. 2025, A. Spencer, pers. comm.).
Resurrect Thryophilus rufalbus sylvus and lump Thryophilus rufalbus transfinis
Summary: (5→5 subspecies)
Details: Clements v2024 taxa 27523–27528
Was:
- Rufous-and-white Wren Thryophilus rufalbus
- subspecies transfinis, rufalbus, castanonotus, cumanensis, and minlosi
Now:
- Rufous-and-white Wren Thryophilus rufalbus
- subspecies rufalbus, castanonotus, sylvus, cumanensis, and minlosi
The former subspecies transfinis (Friedmann, 1947) of southern Mexico (Pacific slope of far southwestern Chiapas) is synonymized with nominate Thryothorus rufalbus rufalbus (de Lafresnaye, 1845), and its range included therein. The formerly synonymized subspecies sylvus Phillips, 1986 is tentatively recognized, with a range (Caribbean slope of southeastern Guatemala and northern Honduras) that was previously unaccounted for in the Clements checklist. However, further study may confirm that sylvus should remain synonymized with the nominate, as by Monroe (1968); see also Zapata (2025).
Resurrect Turdus assimilis suttoni
Summary: (9→9 subspecies, former total including daguae, now split)
Details: Clements v2024 taxa 28724–28734
Was:
- White-throated Thrush Turdus assimilis
- subspecies calliphthongus, lygrus, assimilis, leucauchen, rubicundus, atrotinctus, cnephosus, coibensis, and daguae
Now:
- White-throated Thrush Turdus assimilis
- subspecies suttoni, calliphthongus, lygrus, assimilis, leucauchen, rubicundus, atrotinctus, cnephosus, and coibensis
- Dagua Thrush Turdus daguae (monotypic)
The northeasternmost subspecies suttoni Phillips, 1991, is now recognized by Clements et al. (2025), as it has been in other major global checklists, and now AviList Core Team (2025). In the Clements checklist series, suttoni was previously subsumed within nominate assimilis Cabanis, 1851. See Mlodinow et al. (2025) and Splits section for more details.
Resurrect Hesperiphona vespertina californica and Hesperiphona vespertina warreni
Summary: (3→5 subspecies)
Details: Clements v2024 taxa 32085–32090
Was:
- Coccothraustes vespertinus
- subspecies brooksi, vespertinus, and montanus
Now:
- Hesperiphona vespertina
- subspecies brooksi, californica, warreni, vespertina, and montana
The vocal groups as summarized by Sewell et al. (2004) correspond well with the above subspecies (Gillihan and Byers 2025). These are also now treated as groups (see Changes to Groups). See Generic Changes and Gillihan and Byers (2025) for the change from Coccothraustes to Hesperiphona.
Synonymize eight subspecies of Troglodytes pacificus
Summary: (13→5 subspecies)
Details: Clements v2024 taxa 27288–27294
Was:
- Pacific Wren Troglodytes pacificus
- subspecies alascensis, meligerus, kiskensis, tanagensis, seguamensis, stevensoni, petrophilus, semidiensis, helleri, pacificus, muiri, obscurior, and salebrosus
Now:
- Pacific Wren Troglodytes pacificus
- subspecies alascensis, meligerus, kiskensis, semidiensis, and pacificus
The Pacific Wren Troglodytes pacificus Baird, 1864 has long formed a case in which the number of subspecies recognized seems untenable and unwarranted. While some taxa, mainly insular ones of western Alaska, are clearly distinct, other described taxa differed only inconsistently; see the thorough coverage of this topic in Toews et al. (2025). See also the Changes to Groups section.
Higher-Level Changes
Merge Bucorvidae into Bucerotidae
Summary: (2→1 family)
Details: Clements v2024 taxa 9654–9655
Was:
- Family Bucorvidae
- species Bucorvus abyssinicus and Bucorvus leadbeateri
Now:
- Family Bucerotidae (part)
- species Bucorvus abyssinicus and Bucorvus leadbeateri
Family Bucorvidae has often been recognized for the two species of ground-hornbill Bucorvus Lesson, 1830, as in prior Clements checklists. However, they clearly are members of the same radiation as other hornbills in Bucerotidae, and divergence levels, though evidently deep, are somewhat uncertain and substantially less than for the other families in the Bucerotiformes (Upupidae and Phoeniculidae) (Viseshakul et al. 2011, Gonzalez et al. 2013). Thus AviList Core Team (2025) and Clements et al. (2025) has merged Bucorvidae into Bucerotidae.
Merge Nesospingidae and Spindalidae into Phaenicophilidae
Summary: (3→1 family)
Details: Clements v2024 taxa 33527–33548
Was:
- Family Nesospingidae
- species Puerto Rican Tanager Nesospingus speculiferus
- Family Spindalidae
- species Western Spindalis Spindalis zena, Jamaican Spindalis Spindalis nigricephala, Hispaniolan Spindalis Spindalis dominicensis, and Puerto Rican Spindalis Spindalis portoricensis
- Family Phaenicophilidae
- species Black-crowned Palm-Tanager Phaenicophilus palmarum, Gray-crowned Palm-Tanager Phaenicophilus poliocephalus, White-winged Warbler Xenoligea montana, and Green-tailed Warbler Microligea palustris
Now:
- Family Phaenicophilidae
- species Puerto Rican Tanager Nesospingus speculiferus, Western Spindalis Spindalis zena, Jamaican Spindalis Spindalis nigricephala, Hispaniolan Spindalis Spindalis dominicensis, Puerto Rican Spindalis Spindalis portoricensis, Black-crowned Palm-Tanager Phaenicophilus palmarum, Gray-crowned Palm-Tanager Phaenicophilus poliocephalus, White-winged Warbler Xenoligea montana, and Green-tailed Warbler Microligea palustris
Two Greater Antillean taxa were previously treated as New World warblers Parulidae or incertae sedis after Parulidae: Xenoligea (Chapman, 1917) and Microligea (Cory, 1884), both Hispaniolan, as in Lowery and Monroe (1968, in Volume 14 of Peters’ Check-list). Several other Greater Antillean taxa were treated as tanagers (as by Storer 1970, in Volume 13 of Peters’ Check-list): Nesospingus speculiferus (Lawrence, 1875) of Puerto Rico; Spindalis zena (Linnaeus, 1758), now split into four species occurring throughout the Greater Antilles, including the Bahamas and Cayman Islands; and the two species of Phaenicophilus Strickland, 1851, both limited to Hispaniola. The molecular phylogeny of Barker et al. (2013) showed them to form a clade (and bolstered the case for a breakup of the traditional Tanagers Thraupidae/Thraupinae), but with deep divergences, and was the rationale for the three-family treatment of Chesser et al. (2017) that was adopted by recent Clements checklists.More recently, the UCE phylogeny of Oliveros et al. (2019) recovered all these taxa as monophyletic in most of their reconstructions, and this has led to the merging of Spindalidae and Nesospingidae as a biogeographically cohesive family, though the divergences among these taxa are relatively old (Burns 2025, NACC proposal 2025-C-11, which passed; Chesser et al. 2025). The single-family treatment has also been adopted by AviList Core Team (2025) and Clements et al. (2025).
Sequence Changes
Resequence Tinamous Tinamidae
Details: Clements v2025 taxa 43–230
Was:
- Family Tinamidae
- Nothocercus julius
- Nothocercus bonapartei
- Nothocercus nigrocapillus
- Tinamus tao
- Tinamus solitarius
- Tinamus osgoodi
- Tinamus major
- Tinamus guttatus
- Crypturellus cinereus
- Crypturellus berlepschi
- Crypturellus soui
- Crypturellus ptaritepui
- Crypturellus obsoletus
- Crypturellus undulatus
- Crypturellus transfasciatus
- Crypturellus strigulosus
- Crypturellus duidae
- Crypturellus erythropus
- Crypturellus noctivagus
- Crypturellus atrocapillus
- Crypturellus boucardi
- Crypturellus kerriae
- Crypturellus variegatus
- Crypturellus cinnamomeus
- Crypturellus brevirostris
- Crypturellus bartletti
- Crypturellus parvirostris
- Crypturellus casiquiare
- Crypturellus tataupa
- Rhynchotus rufescens
- Rhynchotus maculicollis
- Nothoprocta taczanowskii
- Nothoprocta ornata
- Nothoprocta perdicaria
- Nothoprocta cinerascens
- Nothoprocta pentlandii
- Nothoprocta curvirostris
- Nothura boraquira
- Nothura minor
- Nothura darwinii
- Nothura maculosa
- Taoniscus nanus
- Eudromia elegans
- Eudromia formosa
- Tinamotis pentlandii
- Tinamotis ingoufi
Now:
- Family Tinamidae
- Tinamotis pentlandii
- Tinamotis ingoufi
- Eudromia formosa
- Eudromia elegans
- Taoniscus nanus
- Nothura boraquira
- Nothura minor
- Nothura darwinii
- Nothura maculosa
- Rhynchotus maculicollis
- Rhynchotus rufescens
- Nothoprocta cinerascens
- Nothoprocta pentlandii
- Nothoprocta curvirostris
- Nothoprocta ornata
- Nothoprocta taczanowskii
- Nothoprocta perdicaria
- Nothocercus julius
- Nothocercus nigrocapillus
- Nothocercus bonapartei
- Tinamus major
- Tinamus solitarius
- Tinamus tao
- Tinamus guttatus
- Tinamus osgoodi
- Crypturellus berlepschi
- Crypturellus cinereus
- Crypturellus ptaritepui
- Crypturellus parvirostris
- Crypturellus tataupa
- Crypturellus obsoletus
- Crypturellus variegatus
- Crypturellus bartletti
- Crypturellus casiquiare
- Crypturellus brevirostris
- Crypturellus soui
- Crypturellus undulatus
- Crypturellus noctivagus
- Crypturellus atrocapillus
- Crypturellus transfasciatus
- Crypturellus cinnamomeus
- Crypturellus boucardi
- Crypturellus kerriae
- Crypturellus erythropus
- Crypturellus duidae
- Crypturellus strigulosus
- Crypturellus [undescribed form]
The linear sequence of Family Tinamidae is reordered based largely on Musher et al. (2024) and Bertelli et al. (2025).
Resequence Cracidae
Details: Clements v2025 taxa 852–975
Was:
- Ortalis vetula
- Ortalis cinereiceps
- Ortalis garrula
- Ortalis ruficauda
- Ortalis erythroptera
- Ortalis wagleri
- Ortalis poliocephala
- Ortalis canicollis
- Ortalis leucogastra
- Ortalis columbiana
- Ortalis guttata
- Ortalis araucuan
- Ortalis squamata
- Ortalis motmot
- Ortalis ruficeps
- Ortalis superciliaris
- Penelope argyrotis
- Penelope barbata
- Penelope ortoni
- Penelope montagnii
- Penelope marail
- Penelope superciliaris
- Penelope dabbenei
- Penelope jacquacu
- Penelope purpurascens
- Penelope perspicax
- Penelope albipennis
- Penelope obscura
- Penelope bridgesi
- Penelope pileata
- Penelope ochrogaster
- Penelope jacucaca
- Pipile pipile
- Pipile cumanensis
- Pipile grayi
- Pipile cujubi
- Pipile jacutinga
- Aburria aburri
- Chamaepetes unicolor
- Chamaepetes goudotii
- Penelopina nigra
- Oreophasis derbianus
- Nothocrax urumutum
- Mitu tomentosum
- Mitu salvini
- Mitu tuberosum
- Mitu mitu
- Pauxi pauxi
- Pauxi koepckeae
- Pauxi unicornis
- Crax rubra
- Crax alberti
- Crax daubentoni
- Crax alector
- Crax globulosa
- Crax fasciolata
- Crax blumenbachii
Now:
- Penelopina nigra
- Chamaepetes unicolor
- Chamaepetes goudotii
- Aburria aburri
- Pipile jacutinga
- Pipile pipile
- Pipile cumanensis
- Pipile grayi
- Pipile cujubi
- Penelope argyrotis
- Penelope bridgesi
- Penelope obscura
- Penelope pileata
- Penelope ochrogaster
- Penelope jacucaca
- Penelope barbata
- Penelope purpurascens
- Penelope superciliaris
- Penelope marail
- Penelope albipennis
- Penelope ortoni
- Penelope montagnii
- Penelope dabbenei
- Penelope jacquacu
- Penelope perspicax
- Oreophasis derbianus
- Nothocrax urumutum
- Pauxi pauxi
- Pauxi koepckeae
- Pauxi unicornis
- Mitu tomentosum
- Mitu mitu
- Mitu salvini
- Mitu tuberosum
- Crax daubentoni
- Crax rubra
- Crax alberti
- Crax globulosa
- Crax blumenbachii
- Crax alector
- Crax fasciolata
- Ortalis vetula
- Ortalis leucogastra
- Ortalis poliocephala
- Ortalis wagleri
- Ortalis cinereiceps
- Ortalis erythroptera
- Ortalis motmot
- Ortalis ruficeps
- Ortalis canicollis
- Ortalis columbiana
- Ortalis guttata
- Ortalis superciliaris
- Ortalis araucuan
- Ortalis squamata
- Ortalis garrula
- Ortalis ruficauda
Linear sequencing of Cracidae now largely follows Hosner et al. (2016).
Resequence Ptilinopus clade of Columbidae
Details: Clements v2025 taxa 2585–2743
Was:
- Genus Ptilinopus
- Ptilinopus cinctus
- Ptilinopus alligator
- Ptilinopus dohertyi
- Ptilinopus porphyreus
- Ptilinopus occipitalis
- Ptilinopus marchei
- Ptilinopus merrilli
- Ptilinopus fischeri
- Ptilinopus jambu
- Ptilinopus epius
- Ptilinopus subgularis
- Ptilinopus mangoliensis
- Ptilinopus leclancheri
- Ptilinopus bernsteinii
- Ptilinopus magnificus
- Ptilinopus perlatus
- Ptilinopus ornatus
- Ptilinopus tannensis
- Ptilinopus aurantiifrons
- Ptilinopus wallacii
- Ptilinopus superbus
- Ptilinopus perousii
- Ptilinopus ponapensis
- Ptilinopus hernsheimi
- Ptilinopus porphyraceus
- Ptilinopus pelewensis
- Ptilinopus rarotongensis
- Ptilinopus roseicapilla
- Ptilinopus regina
- Ptilinopus richardsii
- Ptilinopus chrysogaster
- Ptilinopus purpuratus
- Ptilinopus chalcurus
- Ptilinopus coralensis
- Ptilinopus greyi
- Ptilinopus huttoni
- Ptilinopus dupetithouarsii
- Ptilinopus mercierii
- Ptilinopus insularis
- Ptilinopus coronulatus
- Ptilinopus pulchellus
- Ptilinopus monacha
- Ptilinopus rivoli
- Ptilinopus speciosus
- Ptilinopus solomonensis
- Ptilinopus viridis
- Ptilinopus eugeniae
- Ptilinopus iozonus
- Ptilinopus insolitus
- Ptilinopus hyogastrus
- Ptilinopus granulifrons
- Ptilinopus melanospilus
- Ptilinopus nainus
- Ptilinopus arcanus
- Ptilinopus victor
- Ptilinopus luteovirens
- Ptilinopus layardi
- Ptilinopus [undescribed form]
- Genus Drepanoptila
- Drepanoptila holosericea
- Genus Alectroenas
- Alectroenas nitidissimus
- Alectroenas payandeei
- Alectroenas madagascariensis
- Alectroenas sganzini
- Alectroenas pulcherrimus
- Genus Ptilinopus
- Ptilinopus luteovirens
- Ptilinopus layardi
- Ptilinopus victor
- Ptilinopus arcanus
- Ptilinopus melanospilus
- Ptilinopus holosericea
- Ptilinopus nainus
- Ptilinopus porphyreus
- Ptilinopus alligator
- Ptilinopus cinctus
- Ptilinopus dohertyi
- Ptilinopus superbus
- Ptilinopus rivoli
- Ptilinopus speciosus
- Ptilinopus solomonensis
- Ptilinopus tannensis
- Ptilinopus iozonus
- Ptilinopus eugeniae
- Ptilinopus viridis
- Ptilinopus hyogastrus
- Ptilinopus granulifrons
- Ptilinopus perlatus
- Ptilinopus wallacii
- Ptilinopus aurantiifrons
- Ptilinopus ornatus
- Ptilinopus insolitus
- Ptilinopus pulchellus
- Ptilinopus monacha
- Ptilinopus coronulatus
- Ptilinopus regina
- Ptilinopus mercierii
- Ptilinopus dupetithouarsii
- Ptilinopus greyi
- Ptilinopus porphyraceus
- Ptilinopus pelewensis
- Ptilinopus roseicapilla
- Ptilinopus richardsii
- Ptilinopus perousii
- Ptilinopus ponapensis
- Ptilinopus huttoni
- Ptilinopus insularis
- Ptilinopus coralensis
- Ptilinopus chalcurus
- Ptilinopus rarotongensis
- Ptilinopus chrysogaster
- Ptilinopus hernsheimi
- Ptilinopus purpuratus
- Ptilinopus [undescribed form]
See Generic Changes section, under discussion of Megaloprepia, for details.
Resequence Gruidae (except Balearica)
Details: Clements v2025 taxa 5696–5721
Was:
- Genus Anthropoides
- Anthropoides virgo
- Anthropoides paradiseus
- Genus Bugeranus
- Bugeranus carunculatus
- Genus Leucogeranus
- Leucogeranus leucogeranus
- Genus Antigone
- Antigone canadensis
- Antigone antigone
- Antigone rubicunda
- Antigone vipio
- Genus Grus
- Grus grus
- Grus monacha
- Grus americana
- Grus nigricollis
- Grus japonensis
Now:
- Genus Leucogeranus
- Leucogeranus leucogeranus
- Genus Antigone
- Antigone canadensis
- Antigone vipio
- Antigone rubicunda
- Antigone antigone
- Genus Grus
- Grus carunculatus
- Grus virgo
- Grus paradiseus
- Grus japonensis
- Grus americana
- Grus grus
- Grus nigricollis
- Grus monacha
See Generic Change section, “Subsume Bugeranus and Anthropoides into Grus” for details that required linear resequencing, which follows Krajewsky et al (2010).
Resequence Neopelma and Protopelma
Details: Clements v2025 taxa 15690–15694
Was:
- Genus Neopelma
- Neopelma pallescens
- Neopelma chrysocephalum
- Neopelma aurifrons
- Neopelma chrysolophum
- Neopelma sulphureiventer
Now:
- Genus Neopelma
- Neopelma pallescens
- Neopelma chrysocephalum
- Neopelma aurifrons
- Neopelma sulphureiventer
- Genus Protopelma
- Protopelma chrysolophum
See Generic Change section, “Recognize Protopelma for Neopelma chrysolophum” for rationale for change in linear sequence, which follows van Els et al. (2023).
Resequence genus Turdus
Details: Clements v2025 taxa 28423–28778
Was:
- Turdus mupinensis
- Turdus simensis
- Turdus litsitsirupa
- Turdus viscivorus
- Turdus philomelos
- Turdus tephronotus
- Turdus ludoviciae
- Turdus mandarinus
- Turdus libonyana
- Turdus bewsheri
- Turdus smithi
- Turdus menachensis
- Turdus olivaceus
- Turdus iliacus
- Turdus plebejus
- Turdus lherminieri
- Turdus abyssinicus
- Turdus helleri
- Turdus roehli
- Turdus merula
- Turdus flavipes
- Turdus lawrencii
- Turdus leucomelas
- Turdus fumigatus
- Turdus hauxwelli
- Turdus assimilis
- Turdus albicollis
- Turdus obsoletus
- Turdus rufiventris
- Turdus nudigenis
- Turdus maculirostris
- Turdus sanchezorum
- Turdus grayi
- Turdus haplochrous
- Turdus turdoides
- Turdus infuscatus
- Turdus nigrescens
- Turdus migratorius
- Turdus rufopalliatus
- Turdus rufitorques
- Turdus pelios
- Turdus ravidus
- Turdus aurantius
- Turdus plumbeus
- Turdus xanthorhynchus
- Turdus olivaceofuscus
- Turdus niveiceps
- Turdus swalesi
- Turdus jamaicensis
- Turdus leucops
- Turdus falcklandii
- Turdus reevei
- Turdus subalaris
- Turdus murinus
- Turdus eremita
- Turdus amaurochalinus
- Turdus ignobilis
- Turdus arthuri
- Turdus maranonicus
- Turdus fulviventris
- Turdus olivater
- Turdus nigriceps
- Turdus fuscater
- Turdus serranus
- Turdus chiguanco
- Turdus boulboul
- Turdus simillimus
- Turdus unicolor
- Turdus dissimilis
- Turdus cardis
- Turdus hortulorum
- Turdus pallidus
- Turdus feae
- Turdus obscurus
- Turdus chrysolaus
- Turdus celaenops
- Turdus mindorensis
- Turdus thomassoni
- Turdus nigrorum
- Turdus erythropleurus
- Turdus schlegelii
- Turdus javanicus
- Turdus deningeri
- Turdus papuensis
- Turdus heinrothi
- Turdus bougainvillei
- Turdus kulambangrae
- Turdus vanikorensis
- Turdus pritzbueri
- Turdus xanthopus
- Turdus poliocephalus
- Turdus samoensis
- Turdus ruficeps
- Turdus maximus
- Turdus kessleri
- Turdus pilaris
- Turdus albocinctus
- Turdus rubrocanus
- Turdus torquatus
- Turdus atrogularis
- Turdus ruficollis
- Turdus eunomus
- Turdus naumanni
Now:
- Turdus mupinensis
- Turdus simensis
- Turdus litsitsirupa
- Turdus viscivorus
- Turdus philomelos
- Turdus tephronotus
- Turdus ludoviciae
- Turdus menachensis
- Turdus mandarinus
- Turdus bewsheri
- Turdus libonyana
- Turdus olivaceus
- Turdus abyssinicus
- Turdus helleri
- Turdus roehli
- Turdus xanthorhynchus
- Turdus olivaceofuscus
- Turdus niveiceps
- Turdus boulboul
- Turdus simillimus
- Turdus pelios
- Turdus smithi
- Turdus iliacus
- Turdus merula
- Turdus unicolor
- Turdus dissimilis
- Turdus hortulorum
- Turdus cardis
- Turdus mindorensis
- Turdus thomassoni
- Turdus nigrorum
- Turdus erythropleurus
- Turdus schlegelii
- Turdus javanicus
- Turdus deningeri
- Turdus papuensis
- Turdus heinrothi
- Turdus bougainvillei
- Turdus kulambangrae
- Turdus vanikorensis
- Turdus pritzbueri
- Turdus xanthopus
- Turdus poliocephalus
- Turdus samoensis
- Turdus ruficeps
- Turdus maximus
- Turdus kessleri
- Turdus pallidus
- Turdus obscurus
- Turdus feae
- Turdus celaenops
- Turdus chrysolaus
- Turdus pilaris
- Turdus albocinctus
- Turdus rubrocanus
- Turdus torquatus
- Turdus atrogularis
- Turdus ruficollis
- Turdus eunomus
- Turdus naumanni
- Turdus turdoides
- Turdus ravidus
- Turdus aurantius
- Turdus plumbeus
- Turdus ardosiaceus
- Turdus infuscatus
- Turdus nigrescens
- Turdus rufitorques
- Turdus migratorius
- Turdus rufopalliatus
- Turdus plebejus
- Turdus swalesi
- Turdus jamaicensis
- Turdus leucops
- Turdus lherminieri
- Turdus fulviventris
- Turdus reevei
- Turdus serranus
- Turdus olivater
- Turdus nigriceps
- Turdus fuscater
- Turdus chiguanco
- Turdus falcklandii
- Turdus eremita
- Turdus amaurochalinus
- Turdus ignobilis
- Turdus arthuri
- Turdus maranonicus
- Turdus flavipes
- Turdus subalaris
- Turdus murinus
- Turdus lawrencii
- Turdus obsoletus
- Turdus assimilis
- Turdus daguae
- Turdus albicollis
- Turdus leucomelas
- Turdus hauxwelli
- Turdus fumigatus
- Turdus rufiventris
- Turdus haplochrous
- Turdus nudigenis
- Turdus sanchezorum
- Turdus maculirostris
- Turdus grayi
The genus Turdus, which currently contains 105 species, is resequenced primarily following the phylogenies of Batista et al. (2020) and Reeve et al. (2022, 2023).
